MolStressH2O research cluster uncovers effects of combined global change stressors on Yorkshire’s marine invertebrates Also see the related news item on the University of Hull's website Our oceans suffer from a multitude of stressors. The dramatically rising levels of carbon dioxide lead to warming and acidification, compounded by increasing plastic pollution. Each of these stressors poses a direct threat to aquatic life (such as entanglement or ingestion of plastic debris, or the dissolution of shells through lower pH). But less studied than the direct effects, is how these stressors interact with one another and which consequences such combined effects can have at the molecular level. For example, high temperatures could increase the toxic effect of chemical compounds that leach out of plastics (plastic additives or plasticisers). These chemicals could even be attractive for marine species, leading to ingestion. An indirect effect of ocean acidification is that it can affect the sense of smell, for example by changing the structure (and so the function) of odour molecules sensed by animals. In 2018, the MolStressH2O (molecular stress in changing aquatic environments) research cluster led by Dr. Katharina Valero, PI of OdysysLab (Organismal dynamic systems lab), was formed to study such combined effects at the molecular level. Cluster members have recently published three studies which reveal that marine invertebrates found along the Yorkshire coast (small hermit crabs Pagurus bernhardus and mussels Mytilus spp.), are indeed negatively affected by combined global change-relevant stressors. Plasticiser pollution and global warming will affect male and female mussels differently In a special issue of Marine Pollution Bulletin about multiple stressors in marine ecosystems, MolStressH2O/OdysysLab PhD student Luana Fiorella Mincarelli studied the stress responses of mussels from Filey Bay (Mytilus spp.) exposed to combined end-of-century predicted high temperature and to a plastic softener (di-2-ethylhexyl phthalate, DEHP). DEHP’s use is restricted in several countries including the UK due to its toxic effect on reproduction, but is regardless often found in rivers and seas, and can be viewed as a proxy for other similar but yet unregulated plasticisers. “It is critically important to understand how plastic additives work on molecular levels, especially on reproductive success” - she stated - “their toxic effect can be amplified in a climate change scenario, with biological and breeding repercussions on invertebrates such as mussels, which are commercially-important species worldwide”. In the study, Luana and her team (Prof. Jeanette Rotchell and Dr. Emma Chapman, MolStressH2O/Aquatic Toxicology group, Dr. Alexander Turner, MolStressH2O/Computer Science at the University of Nottingham, and Dr. Katharina Valero, MolStressH2O/OdysysLab) found out that the mussel’s responses to temperature stress and plastic additive exposure varied based on sex: while male mussels were mostly affected by increased temperature, females were more sensitive to DEHP exposure. Specifically, when exposed to high temperature, males accelerated their reproductive cycle and altered their molecular defences against stress. On the other hand, female mussels seemed to be more resilient to climate stressors, but exhibited molecular changes associated with disturbed reproduction when exposed to different concentrations of DEHP. This suggests that a future consisting of higher sea temperatures combined with plasticiser pollution could impact the reproduction and the breeding success of these mussels, by males and females getting “out of synch” with each other. Hermit crabs smell predator odour better under ocean acidification It is commonly expected that ocean acidification impairs the sense of smell of marine animals, thus impeding communication - but such studies typically do not take into account that molecules themselves may change their structure and function under ocean acidification. Challenging our understanding of the inhibiting effects of ocean acidification on the sense of smell, a study led by PhD student Paula Schirrmacher (MolStressH2O/Hull Chemical Ecology group) published in the Journal of Chemical Ecology, however, found an odour molecule that turns out to work better under ocean acidification. Paula and her team (Dr. Christina Roggatz, Energy and Environment Institute, Dr. David Benoit, MolStressH2O/E.A. Milne Centre for Astrophysics and Dr. Jorg Hardege, MolStressH2O/Chemical Ecology Group) dug deeper and combined behavioural choice assays with computational chemistry to explore the function of a chemical cue (2-phenylethylamine, PEA) for hermit crabs from Robin Hood’s Bay in current and end-of-century ocean conditions. The team demonstrated that PEA, which is known to warn mammals and sea lampreys of predators, is actually an attractant for hermit crabs (who are known to be attracted by the smell of death and decay as their food source) and that the potency of this cue increases in an ocean acidification scenario. In order to explain this increased potency, they looked at changes to the structure and charge of PEA, but these were small. “This chemical odour defies all expectations on its function and even turns out to work better under ocean acidification scenarios.”, says Paula and adds: “This challenged our understanding of the mechanisms behind ocean acidification effects on the sense of smell and led to the proof of principle of a completely new mechanistic pathway.” These small changes in the PEA molecule were enough to affect the binding of the PEA molecule to an olfactory receptor. Dr Christina Roggatz, a researcher at Hull whose work features molecule structure changes under ocean acidification, said: “To receive smell-related information correctly, the odour molecule needs to properly fit and bind to the receptor. Our results show how lower environmental pH causes small changes in the chemical properties of the odour molecules, which in turn facilitate this crucial binding step. In short: pH can make or break successful communication in the ocean.” Plastic additive smell attracts hermit crabs Visual similarities of plastics to food as well as odour released from biofouled plastic have been suggested to attract foraging animals - but are they also attracted to the smell of the plastic itself? Dr. Jorg Hardege, member of MolStressH2O and leader of the Chemical Ecology group states: “This is a topic that previously attracted a lot of scientific controversy, but there was not much actual data”. Jack Greenshields, who did his Msc thesis at the Chemical Ecology Hull group and is now a PhD student at Central Queensland University in Australia, together with Paula Schirrmacher, set out to shed light onto this question. They led a study published in Marine Pollution Bulletin showing that hermit crabs are attracted to the plastic additive oleamide at small concentrations in a matter comparable to a feeding stimulant. Furthermore, the hermit crabs’ respiration rate increases significantly when faced with the odour of this slipping agent - an indication that they are excited about what they smell. Interestingly, the chemical structure of oleamide has a striking resemblance to an odour cue known to be involved in chemical communication. As this odour can be naturally released by carrion, scavengers like hermit crabs might interpret the plastic additive oleamide as a feeding cue, potentially creating an olfactory trap. This short communication alerts the scientific community that the abundance of additives leaching from marine plastic debris has the potential to disrupt chemical communication in the oceans, impacting marine food webs and communities.
References: Greenshields, J., Schirrmacher, P. and Hardege, J.D., 2021. Plastic additive oleamide elicits hyperactivity in hermit crabs. Marine Pollution Bulletin, 169, p.112533. https://doi.org/10.1016/j.marpolbul.2021.112533 Mincarelli, L.F., Rotchell, J.M., Chapman, E.C.,Turner, A.P., and Wollenberg Valero, K.C., 2021. Consequences of combined exposure to thermal stress and the plasticiser DEHP in Mytilus spp. differ by sex. Marine Pollution Bulletin, 179, p. 112624. Schirrmacher, P., Roggatz, C.C., Benoit, D.M. and Hardege, J.D., 2021. Ocean Acidification Amplifies the Olfactory Response to 2-Phenylethylamine: Altered Cue Reception as a Mechanistic Pathway? Journal of Chemical Ecology, pp.1-18. https://doi.org/10.1007/s10886-021-01276-9 Intergovernmental Panel on Climate Change (IPCC) (2018). An IPCC Special Report on the impacts of global warming of 1.5° C. Intergovernmental Panel on Climate Change (IPCC) (2014). Climate Change 2014: The physical science basis. Contribution of working group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change.
1 Comment
It takes a global village to understand the effect of climate on lizardsThis blog was first published in Nature Ecology & Evolution "Behind the paper" Our paper “Environmental temperatures shape thermal physiology as well as diversification and genome-wide substitution rates in lizards” published in Nature Communications is about lizards of the family Lacertidae. Many of these love the blazing heat of the Namib or Sahara. But they also have conquered cooler regions of Europe, parts of Northern Africa, and Asia, where high mountain peaks remain glaciated most of the year. Lacertids even include the only reptile whose distribution extends all the way to the North Cape. Dinarolacerta mosorensis, an enigmatic montane endemic of the Balkans. Lizards in temperate regions must bask in the sun to warm up sufficiently to be active. If one does this all day, however, one cannot do other things like foraging or mating or escaping predators. Survival of cold-blooded animals in cold climates certainly requires evolutionary adaptations. Clarifying the species-climate-physiology relationships of lizards would also provide clues to predict their response to a rapidly warming current climate. Thus originated this project. Our question was big, and our lizards are distributed all the way from South Africa to the sub-Arctic. We needed specialists in phylogenomics, physiology and modelling. From the very beginning the project was conceived as teamwork, but we were not expecting that over 40 co-authors from 17 countries would eventually be contributing to this work, with all the opportunities and challenges such a collaboration entails. And in fact, different from many other multi-author collaborations, all of the authors provided very substantial input – so much that determining order of authors became difficult, and our paper now features four authors with equal contribution (Joan Garcia-Porta, Iker Irisarri, Miguel Vences and myself). Emily Darling, after publishing a study with 80 collaborators, has recently written a "Behind the paper" Blog giving recommendations for such big-data research – we had only 45 authors, but some of her tips would have come in handy when we started. We needed to reliably understand the evolutionary relationships between lacertids, which required collecting samples and applying genomic-scale sequencing approaches. To put dates on lacertid evolution we needed numerous novel micro-CT scans of fossils and museum-preserved lizard specimens. No data on preferred temperature were available from many key taxa, so several teams went out into the field to experimentally determine which temperature each species of lizard likes to bask at (by placing them in a tank with a temperature gradient), and how much water they evaporate. To relate physiology to present environment, thousands of distribution records of all species had to be compiled and filtered. Lastly, all the data had to be analyzed on two supercomputers, and integrated. Documenting all data and analyses required altogether 34 supplementary tables and figures. Figure1: Sebastian Kirchhof and Olga Jovanovic "fishing" for lizards in Croatia. Figure 2. Four individuals of Gallotia galloti in a thermal gradient; the specimens have a thermocouple gently attached to their belly, and select their preferred temperature within the gradient. These efforts eventually rewarded us with a reliable time-tree that we could use to address the question how climate influenced the evolution of lacertids. We found that their preferred body temperatures have indeed adapted to their currently occupied climates. Temperate species had low preferred body temperatures, which means they can reach these faster in cold weather, freeing up time for other activities. It surprised us that current environmental temperature experienced by these lizards influenced so many aspects of their biology – geographical species richness, range size, and molecular evolutionary rates were all strongly correlated to temperature. We also solved the conundrum of the high number of lacertid species in temperate climates: their diversity originated predominantly in warm paleoclimates, when presently cold-adapted species remained isolated in geographical refuges and evolved into distinct species. Then, what about climate change? Usually it is thought that tropical lizards are the most threatened. In warm areas, they live under environmental temperatures close to their own thermal preference. If temperatures spike, they quickly become too hot and perish, or they must hide in shaded areas, thus reducing the time they have available for foraging and reproduction. Figure 3. Zootoca vivipara, a the northernmost occurring species of lizard, affected by declines at the southern edge of its distribution.
In contrast, our temperate lizards are commonly thought to be relatively safe as they are assumed to have a larger “thermal safety margin”. With higher temperatures, these sun-loving reptiles would just need to spend less time basking. However, this is clearly not the case – already, numerous lizard extinctions are documented from cool-adapted species (Sinervo et al. 2010). An explanation may be that their tiny distributions on mountains or in shady forests are invaded by stronger generalist competitors. Also, most cold-adapted species have high water loss rates. This makes them vulnerable against drought, which usually comes along with heat. The lower preferred temperature combined with their humidity-handicap means that their love of the sun may not suffice to arm them against a rapidly warming climate with the increasingly frequent heatwaves we already experience. Photo credits: Miguel Vences References: Garcia-Porta et al. Environmental temperatures shape thermal physiology as well as diversification and genome-wide substitution rates in lizards. Nature Communications 10:4077 (2019). Read online at: https://rdcu.be/bQE2n Sinervo, B. et al. Erosion of lizard diversity by climate change and altered thermal niches. Science 328, 894–899 (2010). Genomic Signatures of Climate Adaptation in Anolis cybotesThis blog post was written together with Ariel Rodriguez and first published on Anole Annals Thermal adaptation is the evolution of the ability to persist in novel thermal environments. Phenotypic characters that allow such adaptation, as well as the resulting shifts in the geographic distributions of species, are an emerging field of study in the midst of a changing global climate. Yet, the genomic basis of such phenotypic adaptation is less well understood, so recent efforts of evolutionary biologists are now aiming at one emerging question: Which genes determine thermal adaptation, and are these the same across different populations and species? Luckily, Anolis is yet again at the forefront of novel discoveries being made in this field (see Campbell-Staton et al., 2017). Many studies have independently identified genes that are responding to changes in the thermal environment, be it through change of expression under an acute stress, or through changes in the DNA sequence as evolutionary response. In 2014, we gathered information on such thermal adaptation candidate genes from Drosophila to Homo sapiens from the literature. From the published evidence, we extracted a set of gene functions that potentially underlie climatic adaptation. We were able to match these with functions that are known from phenotypic thermal adaptation (Wollenberg Valero et al., 2014). Interestingly, the products of these genes (Proteins, RNAs) were found to be functionally related with each other thus forming gene networks within the cellular environment. The Caribbean Anolis cybotes is widely distributed across Hispaniola, and thrives in hot, xeric environments just as well as in cooler and more humid montane environments. The rift valley of Lago Enriquillo heats up to 40.5 °C (104.9 °F), and a few instances of frost were reported at the highest peak (Pico Duarte at 3,098m elevation) – so population survival across these climatic extremes does not seem to be a trivial endeavor. Populations of this species show pronounced differences between montane and lowland forms in morphology, physiology, behavior, and perch use (Wollenberg et al., 2013; Muñoz et al., 2014), which led us to expect that at least some of this variation should have a genetic basis. Thus, we set up to test whether Anolis cybotes displays any signatures of genomic adaptation to the diverse kinds of environments it inhabits, and whether any genes showing evidence for selection can also be subsumed under the candidate functions we defined previously. We sampled tissue of these lizards from several high and low elevations (the specimens being the same as in Wollenberg et al., 2013), and looked for variation according to climatic differences via RAD sequencing and subsequent analysis with LFMM. RAD sequencing generates a reduced representation of the target genome, producing thousands of short sequences representing the distribution of the restriction enzyme’s cutting sites throughout the genome. Owing to this property, it cannot be expected that this type of data will necessarily contain “the total set of adaptation genes”; to this effect, detailed genome sequencing is required and such studies have been done in some model organisms (stickleback fish, beech mice, Drosophila, etc.). With our study design, however, we could trace signatures of selection as climate-related changes in the allelic frequencies in the fragments that were sequenced. We identified a total of 84 SNPs with statistical signatures of selection and 14 of these matched protein-coding genes on different chromosomes of the Anolis carolinensis genome (the best available reference). Not surprisingly, our data set and analysis did not “hit” any major known candidate genes for thermal adaptation, but we made another discovery. Most of the genes that we did identify as having adapted to the different climatic environments in Anolis cybotes populations perform the set of previously predicted gene functions that we had predicted in 2014 (Rodríguez et al., 2017).
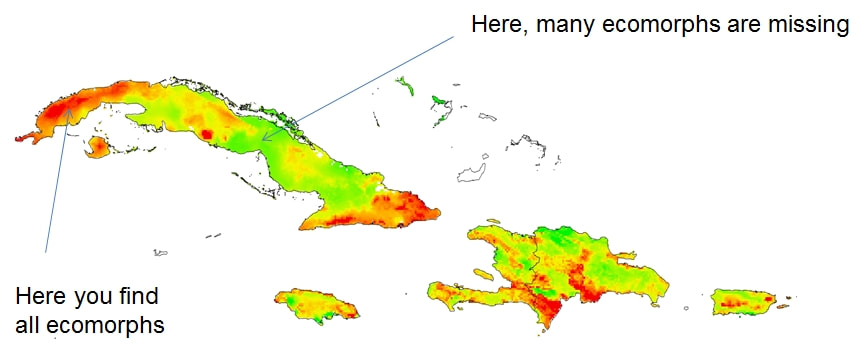
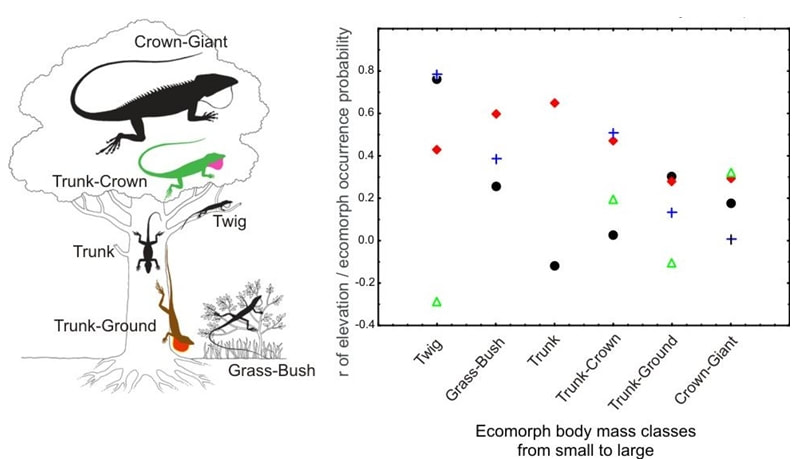
Moreover, the newly identified genes were also in close functional connection with each other, and with many of the previously predicted genes, forming a tightly knit functional network. Some forays into tissue expression databases further revealed that many of these genes are also expressed in brain, and during early development. The figure shows candidate gene functions for thermal adaptation that we predicted in 2014 and now verified in Anolis cybotes Due to the fact that we couldn’t scan the entire genome of Anolis cybotes (so far unknown), we may have missed part of the story. However, it is encouraging that at least one of the genes adapting to different climatic environments in the green anole, A. carolinensis, also has one of the functions we predicted (vasodilation, constriction and regulation of blood pressure, Campbell-Staton et al., 2016). Phenotypic adaptation to climate can happen in several different ways and we are yet only scratching the surface of the genetic basis of this phenomenon. For reptiles, their thermoregulatory behavior, water balance in hot climates, freeze tolerance, and anti-oxidative strategies seem to be the most important eco-physiological challenges (Storey and Storey, 2017), and we deem it likely that changes in a multitude of genes are contributing to these adaptive responses. Our study provides evidence that climate adaptation on the genomic level is constrained to specific organismal functions and biochemical pathways, which may underpin the observed molecular and phenotypic differences. The definitive answer to the question on whether the same set of genes underlie climatic adaptation across populations and species, is yet to be found; but the study of the functional connections between genes can be very informative for this endeavor. You can read our full paper here (open access) what makes anolis ecomorph communities complete?This blog post was originally published on Anole Annals: One of my favorite graphic representations of a typical anole community is the one where all ecomorphs are hanging out together in a tree and a scrub next to said tree. Each ecomorph has its structural microhabitat place and they are all spaced out evenly across the tree to represent competition. Originally the figure was published by Williams (1983) and then modified later on. Arriving on the Greater Antilles, one thus expects to promptly be able to say hi to all these ecomorphs at the next best tree. Well, from my personal experience, I can tell you that this is unfortunately not the case. Localities where all ecomorphs are found together are scarce, and all of them are famous, having served as field sites for the most ground-breaking of anole discoveries. But what about the rest of them? Something must prevent the co-occurrence of ecomorphs in all these other places. This was noted before: Losos (2009) remarked that all utilized structural microhabitats exploited by all ecomorphs are present throughout the islands, so “complete” ecomorph communities should also be able to occur everywhere. A common explanation for the absence of certain “functional types” (= Anolis ecomorphs) from local communities is a process that is called “filtering.” Modern community assembly theory distinguishes two such types of filters: 1. Biotic interaction filters and 2. Environmental filters. Biotic filtering involves competitive exclusion: For anoles this phenomenon caused ecological speciation which led to the convergent evolution of the ecomorph communities. But biotic filtering should not be expected to occur at this stage of the radiation: Different ecomorphs are not competing for the same structural microhabitat niche in different localities. This leaves environmental filtering. In our study published in Ecology and Evolution, on which I am reporting here, we tested whether environmental filtering could be a possible explanation for the absence of ecomorphs in local communities. First, we modelled Anolis ecomorph community completeness by constructing environmental niche models for each ecomorph (the sum of species belonging to that ecomorph) on each island. These models were then overlaid for all ecomorphs per island. The map for ecomorph community completeness shows a very patchy distribution of areas where all ecomorphs are expected to occur. Comparisons of environmental niches among these islands revealed that only Hispaniola and Cuba have their complete Anolis ecomorph communities occurring in a similar bioclimatic parameter space. This patchiness could be explained by elevation for all islands except Jamaica: the Anolis community completeness map strongly resembles the topographic relief of the Greater Antilles. Looking more closely into the climatic parameters, Jamaica has much lower daily and annual temperature ranges which are also not related to the island’s elevation, whereas in the rest of the Greater Antillean islands, they are. Occurrence probability of ecomorphs seems to be coupled to environmental parameters, which explains why some ecomorphs are “filtered out“ in some locations: they do not encounter a favorable environment there. Since I mentioned initially that filtering relates to “functional types” (not species), the filtering must be a result of certain functional properties of the Anolis ecomorphs’ phenotype. We wanted to take the study a step further and actually investigate one (among many) possible functional trait: body mass. Body mass is a known functional trait in animals and has been found to explain metabolic rate variation in squamate reptiles which is in turn possibly related to thermal physiology. Thermal physiology could determine the parameters of an animal’s functional environmental niche Also, Anolis ecomorphs strongly vary in body mass. In this paper, we identified a significant correlation between environmental filtering of Anolis ecomorph community assembly and ecomorph body mass as a functional trait: Smaller ecomorphs with lower body mass are restricted to higher elevations (where it is colder), while larger ecomorphs are found toward warmer environments (usually found in lower elevations). The Jamaican twig ecomorph Anolis valencienni was the only species included in our study that did not follow the overall pattern.
In conclusion, our results in Anolis expand the understanding of community assembly within insular adaptive radiations of animals. The current consensus is that biotic filters are of importance for their evolution, while environmental filters mostly influence species composition. Here, we could show that environmental filtering of functional traits independently from species composition is a major determinant for local community assembly, and future research should be directed toward investigating the importance of these filters in other adaptive animal radiations. You can see the full paper here (Open Access): http://onlinelibrary.wiley.com/doi/10.1002/ece3.908/abstract;jsessionid=8A448C88B0149BAF4244B42D098A2765.f04t02 Two souls, alas, are dwelling in the amphibian breast
Tadpoles and frogs evolve as two separate life forms – despite being only oneChildren worldwide are mesmerized by the amphibian life cycle as example for the complexity of life forms – An egg turns into a tadpole which then undergoes metamorphosis to develop into a frog. But why is this step necessary? Couldn’t the tadpole have “learned” to reproduce, not needing the frog anymore? Or why don’t more frogs directly develop from eggs, foregoing the tadpole? – Ergo, what’s the evolutionary advantage of having both? Charles Darwin was already puzzled by this question, looking at insect larvae and adult insects who likewise undergo metamorphosis. Since they are part of the same life form, he assumed that evolution in one phase surely must be mirrored by the other. But, since there are also such striking differences between a larva and an adult after metamorphosis, maybe evolution affects both phases differently, driving them apart? From this standpoint, one might invoke Goethe’s Faust, reciting: “Two souls, alas, are dwelling in my breast /And one is striving to forsake its brother”. To decide between these “Darwinian” and “Faustian” viewpoints, an international team of researchers led by the University of Hull and Technical University of Braunschweig studied and compared frog and tadpole evolution of the model frog Xenopus, and the Madagascan mantellid frogs who encompass over 400 species descending from a common ancestor. In the study published in Nature Communications on May 15th, 2017, they found that the evolution of tadpoles is entirely independent from the evolution of frogs, despite that they merely represent two phases of the same organism — Faust: 1, Darwin: 0. “Tadpoles and frogs evolving independently from one another is the explanation for the presence of two such strikingly different phases in the first place”, says Katharina Wollenberg Valero from the University of Hull. “Having different genetic programs responsible for generating adult and tadpole forms may allow each phase to adapt to changes in their environment independently, bypassing possible ill consequences for the other phase”. Publication Wollenberg Valero, K. C., J. Garcia-Porta. A. Rodriguez, M. M. Arias Villarraga, A. Shah., R. D. Randrianiaina, J. L. Brown, F. Glaw, F. Amat, S. Kunzel, R. D. Isokpehi, D. Metzler, and M. Vences. Phenotypic evolution is uncoupled among frog life history phases. Nature Communications, DOI:10.1038/NCOMMS15213. Contact and inquiries: Katharina Wollenberg Valero: [email protected] Miguel Vences: m.vences-tu-bs.de Original blog: Dieser Beitrag wurde am 18. Mai 2017 von Stefan Schmidt unter ZSM veröffentlicht. |
Details
Kat / other membersWe'll update you on news from our research and other related activities Archives
March 2022
Categories |




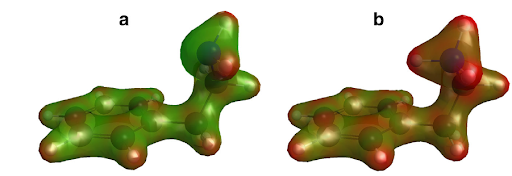








 RSS Feed
RSS Feed
